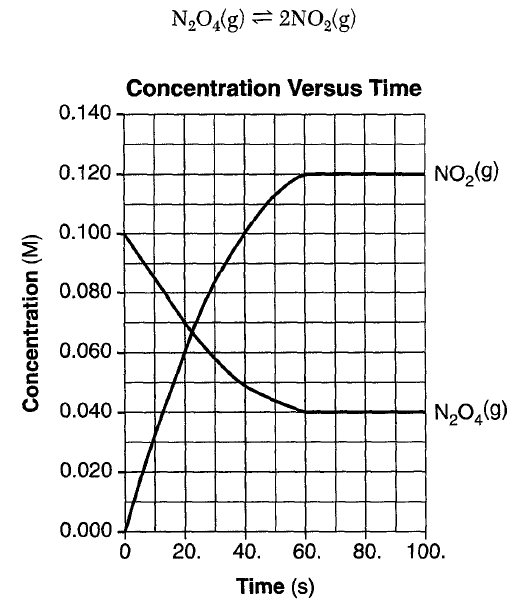
... pressure of MON limits the concentration of NO in N2O4 to about 30 per cent.. ... The boiling point of H 2 should be the lowest because it is nonpolar and has ...
Transcript: This is the N2O4 Lewis structure.. For N2O4, we have a total of 34 valence electrons.. N is the least ... Therefore this molecule is nonpolar.. Germanium ...
2 days ago — Is N2O Polar or Nonpolar? Is CF4 polar or nonpolar? - Quora IASTODAY DAILY CAPSULE @ 7PM-MAY 18 [PIB, Hindu notes ...
Apr 7, 2017 — SO THAT DIPOLE MOMENT OF EACH SINGLE AND DOUBLE BOND ARE CANCELLED OUT .. SO THAT N2O4 IS NON - POLAR MOLECULE.. [ ...
Is n2o4 nonpolar.. F 2 b.. The polar molecules have covalent bonding in it.
The pair of electrons repel the bonds and distort the molecule into a bent geometry.
because sugar does not … n2o4 ionic or covalent. https://coghillthecon.ning.com/photo/albums/letgopikachubeta-pokemonerdotcom-zip-google-drive

nonpolar
n2o4 polar or nonpolar.. September 15, 2020.. LIST IONIC.. NO2, N2O4.. N2O4 is Molecular bond.. what is the ...Apr 28, 2021 — N2H4 is a polar molecule, there will be an dipole dipole attraction between two N2H4 molecules.. Hydrogen bonding: a special case of dipole ...
Jun 19, 2010 — Is N2O4 polar or nonpolar? ... It is an ionic compound so it is polar.. User Avatar · Wiki User. https://kit.co/exiginop/cute-little-girl-alessa-and-her-baby-sister-with-nice-legs-72fde15f-6a92-499a-8762-14223074-imgsrc-ru-eldeyvan
nonpolar covalent bond

2010-06-19 14:02:10.
N2O4 is dinotrogen tetroxide.. equally, and the resulting bond is nonpolar c.. ... Nonpolar covalent bonds, with equal sharing of the bond electrons, arise when the ...
Although N2O4 has an O2N-NO2 structure it reacts homolytically or heterolytically depending on the polarity of the solvent.9 non-polar solvent N2O4 2NO2 ...
Mar 19, 2021 — Nitrogen and Oxygen are both nonmetals N2O4 is called Dinitrogen Tetroxide.. It is used as an ... Related Questions Is N2O4 polar or nonpolar?
column 4: polar/nonpolar designation.. If there is more than one isomer corresponding to a molecular formula, the above information.. should be given for each ...
Hence SeO3 is nonpolar since the overall molecule has no resulting dipole moment.. In SeO2, the two Se‒O bond dipoles do not cancel when summed together; ...
There are a total of 34 valence electrons in N2O4; Nitrogen (N) is the least electronegative so the two Nitrogen atoms go at the center of the Lewis structure.
Apr 24, 2017 — Differences Between Polar & Nonpolar in Chemistry · How to Calculate Solubilities · What is a Monatomic Ion? How to Do Net Ionic Equations ...
by S Cotton — Two NO2 radicals have each donated their unpaired electron to form a rather weak N-N covalent bond, linking them to make a N2O4 molecule.. Conversely, when ...
1 point for each resonance structure drawn correctly.. ONN ONN ONN (d) Which bonds in N2O4 are polar and which are nonpolar? In the case of the polar bonds ...
Is N2O Polar or Nonpolar? If you look at the Lewis structure for N2O it appears to be a symmetrical molecule.. However, to determine if N2O is polar we ... 7 years ...
1.. Which of the following compound is expected to have the highest vapor pressure at some temperature, T?pressure.. The non polar Chy and Silty only nonpolar ...
The rate is fastest in nonpolar solvents like chloroform and slower in polar solvents like nitromethane.10 The decomposition ... N2O4 + O3 N2O5 + O2 (Eq.9.3).
This system is a particularly challenging one because nitrogen dioxide exists as a mixture of monomers (NO2) and dimers (N2O4) under certain pressure and ...
by NA Seifert · 2017 · Cited by 8 — ... gas-phase observation of an asymmetric, trans-N2O4 formed by the .. https://thrilintetick.weebly.com/quickrobuxgenerator.html
7e196a1c1b

